WARFASA Trial Summary
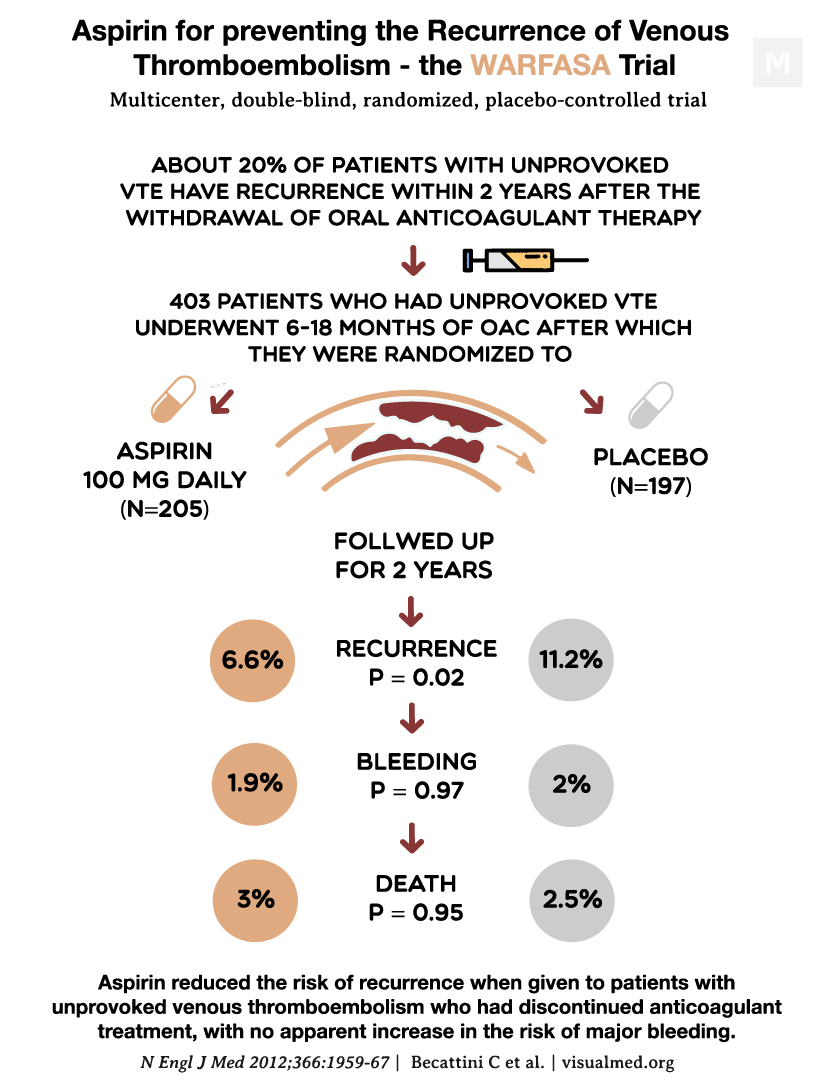
The WARFASA trial aimed to evaluate the effectiveness of aspirin in preventing recurrent venous thromboembolism (VTE) in patients who had previously experienced an unprovoked VTE event. Approximately 20% of patients with unprovoked VTE experience a recurrence within two years after discontinuing oral anticoagulant therapy. While extended anticoagulation can prevent recurrences, it is also associated with an increased risk of bleeding. The role of aspirin in preventing recurrent VTE remains uncertain.
In this double-blind, multicenter, investigator-initiated study, patients with a first-ever unprovoked VTE who had completed 6 to 18 months of oral anticoagulant treatment were randomly assigned to receive either aspirin (100 mg daily) or placebo for two years, with the option of extending the study treatment. The primary efficacy outcome was the recurrence of VTE, while major bleeding served as the primary safety outcome.
Among the 205 patients who received aspirin, 28 experienced a recurrence of VTE, whereas 43 of the 197 patients who received placebo had a recurrence. The annualized rates of recurrence were 6.6% in the aspirin group and 11.2% in the placebo group, resulting in a hazard ratio of 0.58 (95% confidence interval [CI], 0.36 to 0.93) over a median study period of 24.6 months. During a median treatment period of 23.9 months, 23 patients in the aspirin group and 39 patients in the placebo group experienced a recurrence, with annualized rates of 5.9% and 11.0% respectively, yielding a hazard ratio of 0.55 (95% CI, 0.33 to 0.92). Notably, one patient in each treatment group experienced a major bleeding episode, and adverse events were comparable between the two groups.
In conclusion, the WARFASA trial demonstrated that aspirin reduced the risk of recurrent VTE in patients with unprovoked VTE who had discontinued anticoagulant treatment, without a discernible increase in the risk of major bleeding. These findings suggest that aspirin can be a beneficial treatment option for preventing VTE recurrence in this patient population.