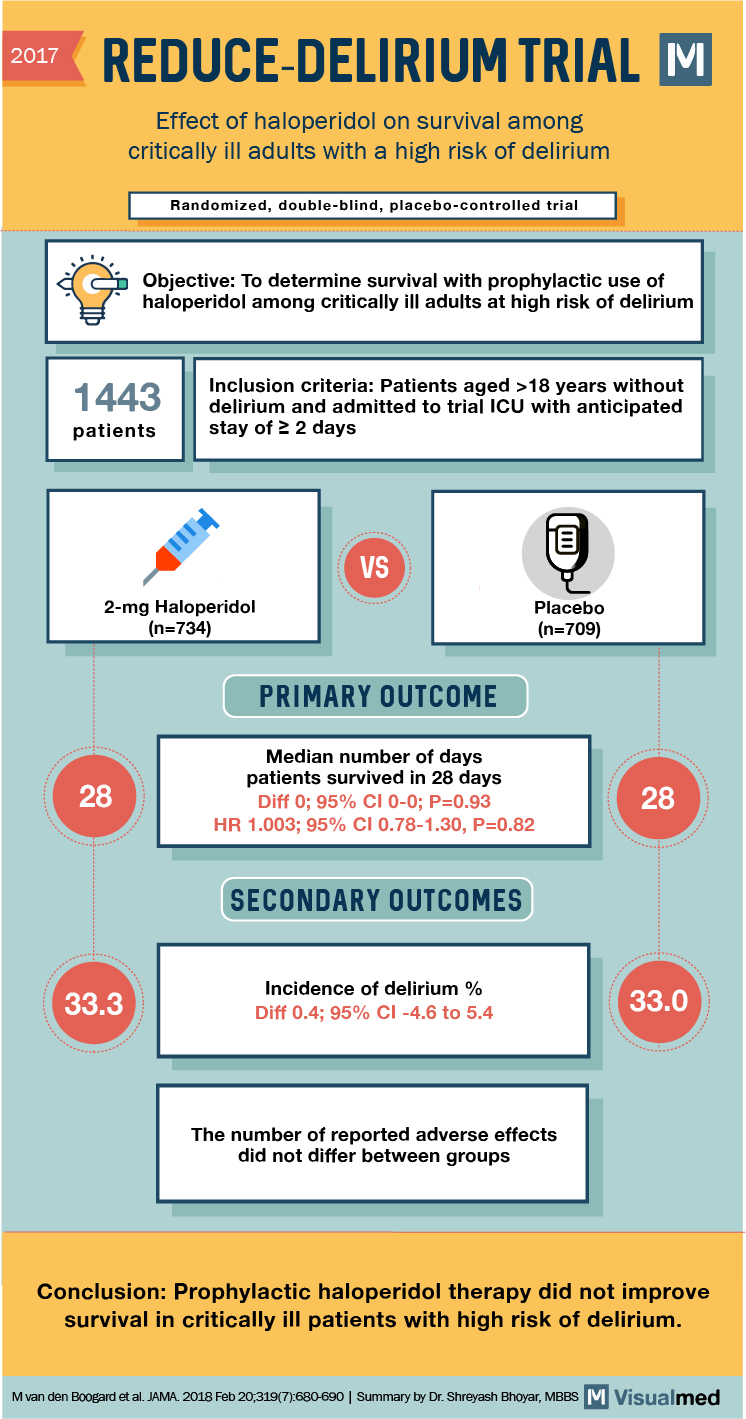
2017 2017 REDUCE-DELIRIUM TRIAL M Effect of haloperidol on survival among critically ill adults with a high risk of delirium | Randomized, double-blind, placebo-controlled trial Objective: To determine survival with prophylactic use of haloperidol among critically ill adults at high risk of delirium 1443 patients Inclusion criteria: Patients aged > 18 years without delirium and admitted to trial ICU with an anticipated stay of 2 days 2-mg Haloperidol (n=734) Placebo (n=709) PRIMARY OUTCOME Median number of days patients survived in 28 days Diff 0; 95% CI 0-0; P=0.93 HR 1.003; 95% CI 0.78-1.30, P=0.82 SECONDARY OUTCOMES 33.3 Incidence of delirium % Diff 0.4; 95% CI-4.6 to 5.4 33.0 The number of reported adverse effects did not differ between groups Conclusion: Prophylactic haloperidol therapy did not improve survival in critically ill patients with high risk of delirium. M van den Boogard et al. JAMA. 2018 Feb 20,319(7):680-690